|
This past year has proven once again that in vivo imaging is an important aspect in the microCT world. Whether it is to image the lungs with or without lung pathologies, for longitudinal studies in mice or rats, one of the most important parts is following up of the animal welfare while scanning. Bruker’s in vivo microCTs have an integrated monitoring system, no third-party software or hardware is necessary. What’s more, the user and animal friendly set-up ensure minimal stress on both parties. This new MN 139 will help you remember how to use the monitoring system or give you the info you need to get started!
As mentioned before, the monitoring system is integrated into the in vivo microCT scanners. The hardware consists of a visual camera with real time body movement detection, ECG connections with sensitive amplifier and pads, and an airflow sensor for direct breathing detection. An ambient temperature sensor and warm air heating are also included to create a complete safe and cozy environment for the anesthetized animal.
Raw image - in XY view
 Figure 1. All connections and sensors for the animal bed of the in vivo SKYSCAN systems All the physiological information is real time gathered and send to the workstation where the user can continuously monitor the animal using an integrated program, aptly named SKYSCAN Visual. Additionally, time marks of the physiological processes can be recorded, effectively using the sensors for capturing the breathingrate and/or heartrate during the scanning process.
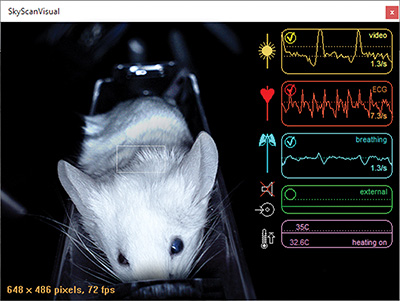
Figure 2. Monitoring window SKYSCAN Visual – A mouse is positioned in the view of the visual monitoring camera and its breathing (on video and with the breathing sensor) and cardiac rhythm (with ECG clips) are being measured.
The breathing of the animal can be followed up in two different ways, either using the visual camera when under inhalation anesthesia or the breathing sensor when using injection anesthesia. Both prove effective in making sure that the depth of the anesthesia is not to deep or to light. Additionally, the connection for the use of an external probe, such as an external ventilator, is also available as standard.
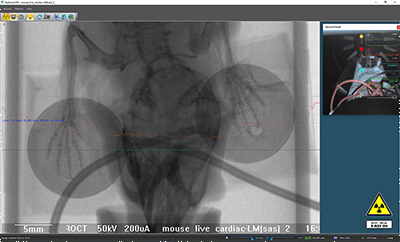
Figure 3. Top view of the transmission through the mouse and the ECG pads, none of the parts contain any high dense parts. The heartrate can be measured using the ECG electrodes or pads. These sticky pads adhere to the paws of the animal and contain no metal, so they don’t produce artefacts.
The ambient temperature in the animal bed can also be set and kept stable to minimize the stress of the anesthesia. Finally, the in vivo scanners have an opening on the side of their animal bed housing to give access to additional wiring, tubing… which might be necessary for your experiment. |